
The standard for de-identification of DICOM objects is defined by the DICOM Standard PS 3.15-2011 Digital Imaging and Communications in Medicine (DICOM), Part 15: Security and System Management Profiles. Examples: Study Instance UID, SOP Class UID, SOP Instance UID. The uses of such UIDs are defined in the various Parts of the DICOM Standard. 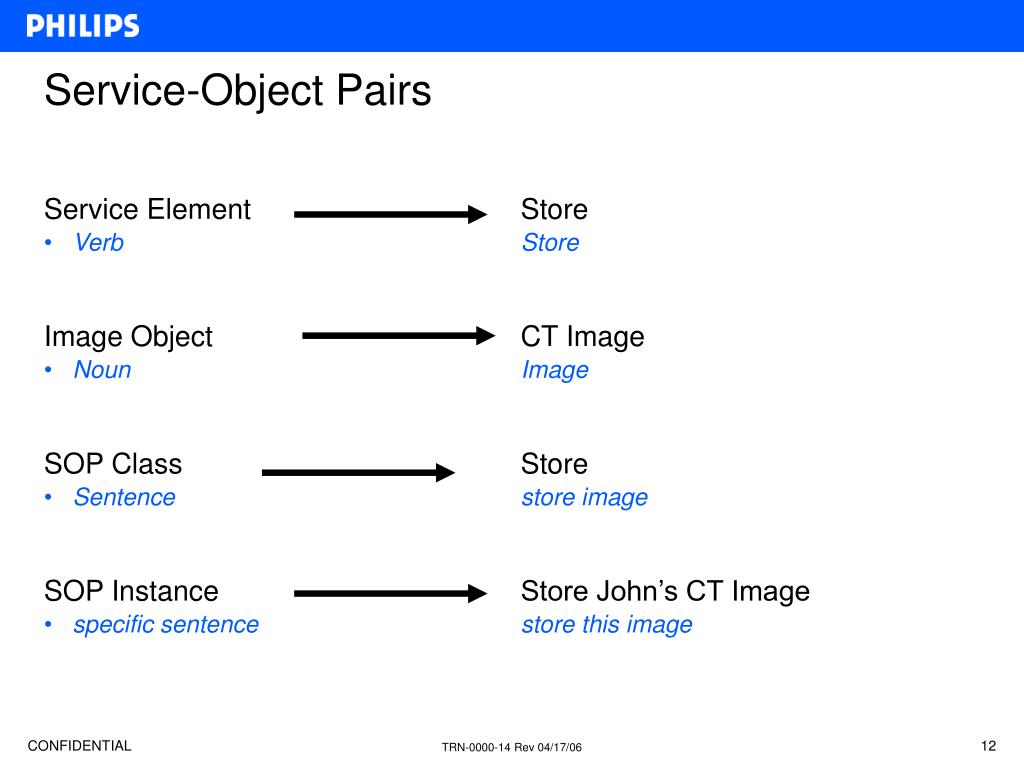
All Unique Identifiers, used within the context of the DICOM Standard, are registered values as defined by ISO 9834-1 to ensure global uniqueness. These requirements are defined the Health Insurance Portability and Accountability Act (HIPAA) section 164.514(b)(2) of the HIPPA Privacy Rule. Name, Patient ID, Patient Birth Date, and Patient Sex. The UID identification scheme is based on the OSI Object Identification (numeric form) as defined by the ISO 8824 standard. The de-identification process is developed in accordance with the requirements set forth by the Federal Drug Administration (FDA) and the European Medicines Agency (EMA). Introduction to patient de-identificationįollowing industry best-practices, the cloud-platform uses a standards-based approach to de-identification of DICOM images to insure that images are free of protected health information (PHI). RECOMIA maintains a license to the SliceVault cloud-platform, and as data sub-processor SliceVault acts only upon work-instructions given by RECOMIA. The cloud platform made available to RECOMIA and its research partners is delivered and maintained by SliceVault, a company dedicated to providing secure and compliant cloud-based solutions for managing medical images in clinical research.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
